 
At the right end, elements whose number of protons is a magic number are lined up corresponding to the rare gas. Therefore, by selecting the most representative isotopes for each element, we devised a periodic table of nuclei corresponding to the periodic table of the elements introduced in Fig. A nuclear chart that covers all of them is known, but the correspondence with the periodic table of elements is not visible. The number of neutrons also greatly affects the stability of the nucleus. Then, is it possible to make a periodic table of the nucleus? Since the nucleus contains neutrons in addition to protons, there are isotopes with different numbers of neutrons even for the same element. Neutrons have a similar magic number, which is the same as protons, except that it is 126 instead of 114. Also, when the number of protons reaches 40, it shows similar properties to a lesser extent. In the case of atoms, the number of electrons is 2, 10, 18, 36, 54, 86, but in the case of atomic nuclei, the number of protons is 2, 8, 50, 82, It's time to reach 114. 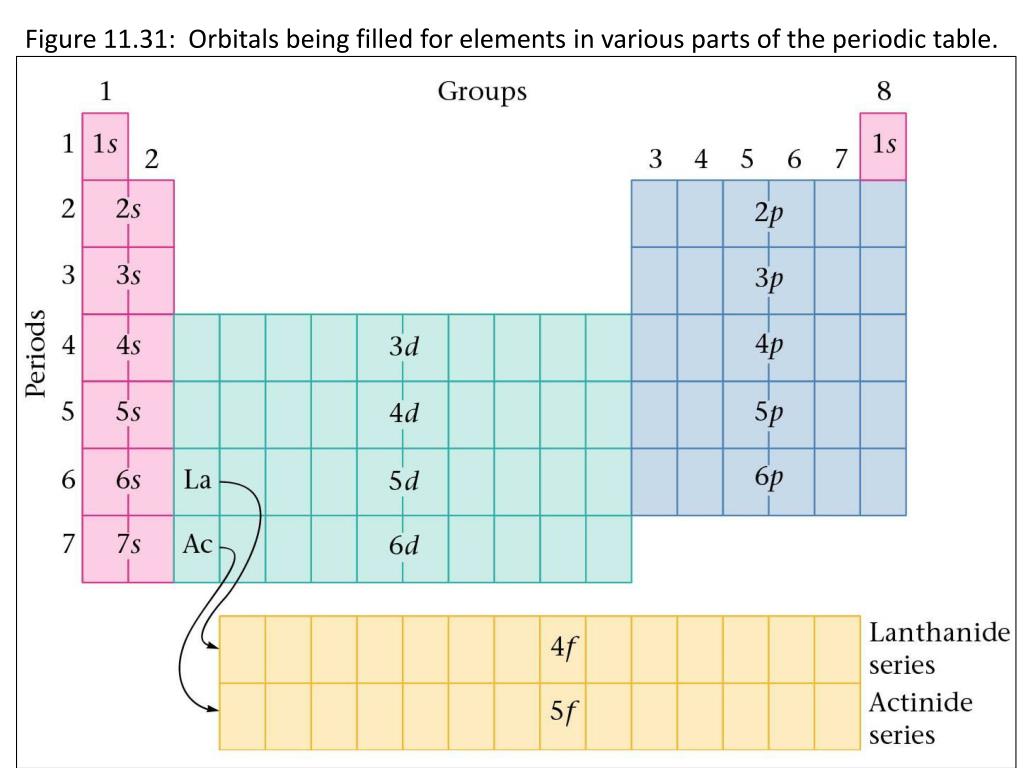
2, the maximum number of particles that each shell can accommodate differs greatly between the case of atoms and the case of atomic nuclei. However, since the environment (potential) of electrons and protons / neutrons is different as shown in Fig. The magic number nucleus becomes a nucleus whose shell is just buried, and corresponds to rare gas. 
This is called the nuclear shell model (Karamokei, shell model) (Note). However, due to the interaction that acts on protons and neutrons (strong interaction called "nuclear force" that Hideki Yukawa considered), orbits that are divided into several shells like electrons are formed, and the orbits are clogged in order from the lowest energy orbit. There is no other "nucleus" at the center of the nucleus. In fact, we can think of similar orbital motions for the protons and neutrons that make up the nucleus itself. in the rightmost column are called rare gases, and because the electrons have just filled the shell of the orbit, they are very stable elements that do not cause a chemical reaction. The colors in Figure 3 show the difference in orbit. The Periodic Table of the Elements expresses how the electrons that go around it are clogged in order from the orbit with the lowest energy. The nucleus has positive electricity proportional to the number of protons. Periodic Table of the Elements (Werner's Periodic Table of the Period) *Click the image to see a larger version.įigure 3. This achievement was published in the academic journal Foundations of Chemistry (April 21, 2020) published by Springer Nature. This new periodic table was devised by Professor Koichi Hagino and Yoshiteru Maeno of the Graduate School of Science, Kyoto University. Surprisingly, however, I haven't found an example of it in the form of a periodic table. It is well known that the nucleus becomes stable when the number of protons contained in the nucleus becomes 2, 8, 20, 28. Are lined up.Īctually, this is the periodic table of "nuclei". The order of Sui, He, Lee, Be (Hydrogen H, Helium He, Lithium Li, Beryllium Be) is the same, but under helium, oxygen, calcium, etc., instead of the rare gas stabilizing elements neon, argon, etc. 
It is similar to the periodic table of elements that appears in junior high school textbooks, but the arrangement of element symbols is different. Periodic Table of Atomic Nuclei "Nucletouch" Overviewįirst look at the new periodic table below. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
